An azeotrope is indicated by the composition at which the isotherm becomes vertical. Points between the saturated liquid line and the saturated vapor line represent a two-phase, liquid-vapor system. Temperature is represented by isothermal tie lines between the saturated liquid (boiling) line and the saturated vapor (dew) line. Composition – Ponchon-Savarit Plot 3 phases are shown on the plot – solid, liquid, and vapor. Composition – Ponchon-Savarit PlotĮnthalpy vs. composition plot to obtain this information. We will also need to employ energy balances, based on enthalpy, for certain separation problems. Composition – Ponchon-Savarit Plot We have begun to employ mass balances, both total and component. The diagram below is based on the vapor-liquid equilibrium of a hypothetical binary liquid mixture and illustrates how an increase in either the pressure or temperature decreases the relative volatility of the mixture.Enthalpy vs.
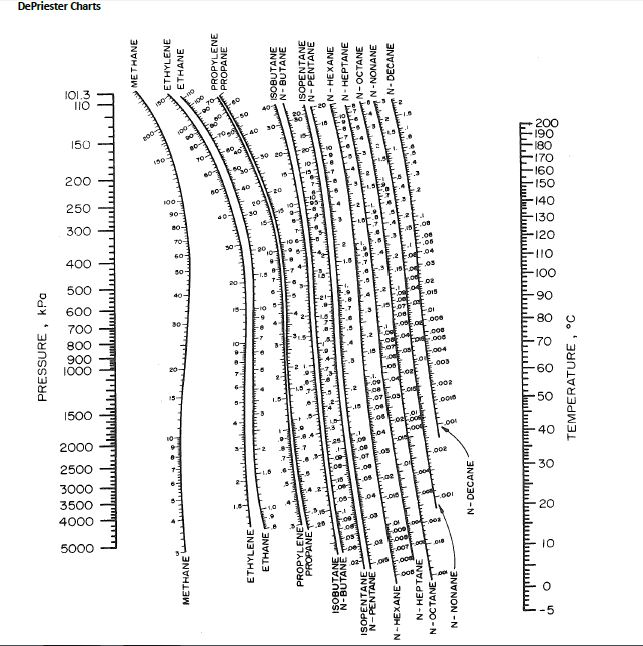
Since an increase in the pressure requires an increase in the temperature, then an increase in temperature also effects the relative volatility. Values are widely used in the design of large-scale distillation columns for distilling multi-component mixtures in petroleum refineries, petrochemical and chemical plants, natural gas processing plants and other industries.Įffect of temperature or pressure on relative volatilityĪn increase in pressure has a significant effect on the relative volatility of the components in a liquid mixture. The values of have been correlated empirically or theoretically in terms of temperature, pressure and phase compositions in the form of equations, tables or graph such as the well-known DePriester charts. Large-scale industrial distillation is rarely undertaken if the relative volatility is less than 1.05. Thus, for the distillation of any multi-component mixture, the relative volatility is often defined as In that context, a lighter component means a component with a lower boiling point (or a higher vapor pressure) and a heavier component means a component with a higher boiling point (or a lower vapor pressure). The column designer would designate the key components governing the separation design to be propane as the so-called light key (LK) and isobutane as the so-called heavy key (HK). Such a distillation column is typically called a depropanizer.
When the volatilities of both key components are equal, it follows that = 1 and separation of the two by distillation would be impossible under the given conditions. That means that ≥ 1 since the larger value of the more volatile component is in the numerator and the smaller of the less volatile component is in the denominator. Thus, a value (= ) for a more volatile component is larger than a value for a less volatile component. When their liquid concentrations are equal, more volatile components have higher vapor pressures than less volatile components. = commonly called the K value or vapor-liquid distribution ratio of a component = the vapor-liquid equilibrium concentration of component in the liquid phase = the vapor-liquid equilibrium concentration of component in the vapor phase = the relative volatility of the more volatile component to the less volatile component

Schematic diagram of a large-scale binary distillation column.įor a liquid mixture of two components (called a binary mixture) at a given temperature and pressure, the relative volatility is defined as
